Are you looking to partner with a trustworthy clinical trials site?
Neuroaxis Research has the expertise, site staff team, logistics and patient pool to support drug, medical device, diagnostic and combination product research programs at the highest standards.


Good Healthcare
At All Times.
Whether you are looking to carry out a single study or embark on a challenging and extensive clinical trial, you can trust Neuroaxis Research to supply optimal site planning, investigators, clinical personnel, participants and data quality.
The right site.
The right investigators.
The right patients for your clinical trials.
Led by an experienced team,
Neuroaxis Research is
equipped to conduct high-quality,
predictable, and efficient clinical
trials in different healthcare sectors.
Keep reading to learn how our
unique model ensures operational
excellence for every trial.

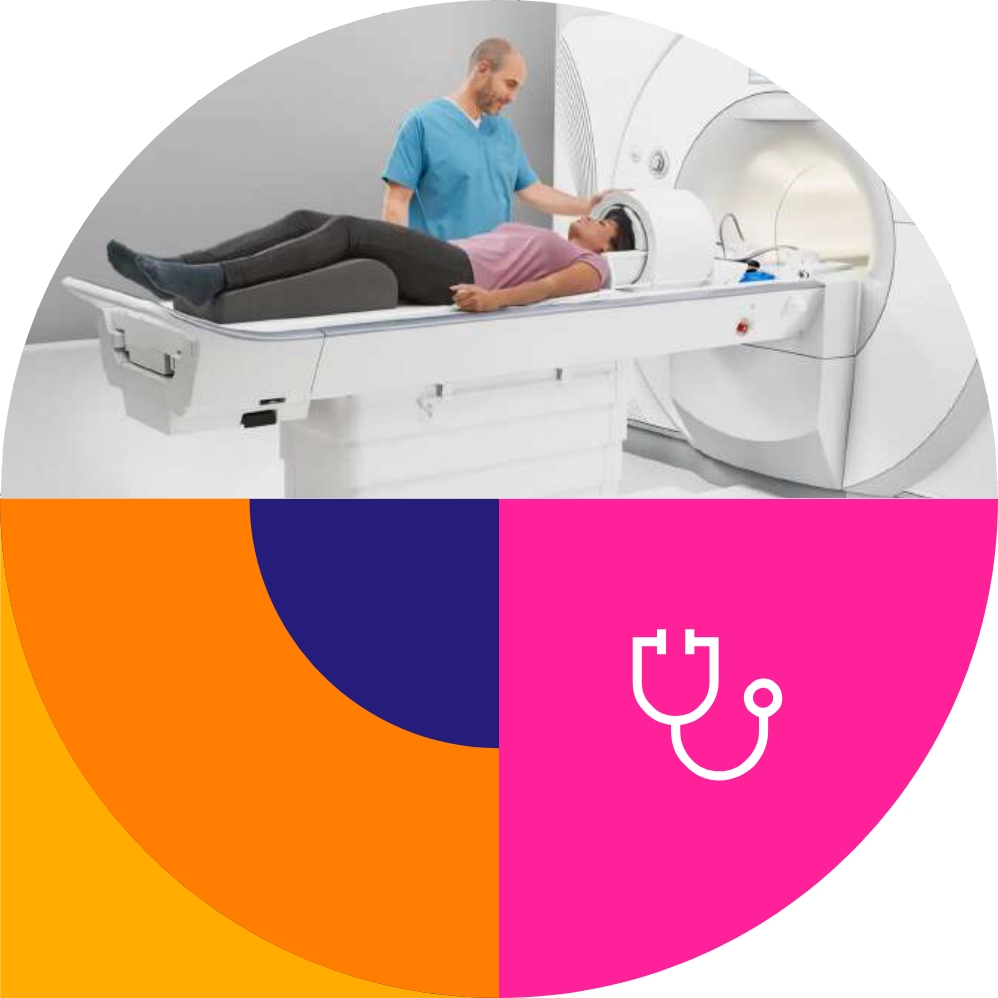
Extensive on-site resources
- Top location & medical facilities
Neuroaxis Clinic is a modern medical location, easy to reach from Bucharest center. Designed by top medical architects, it offers a purpose-built, dedicated, and flexible space for research. Equipped with top medical technology, the space adapts to accommodate specific study logistics and meets the diverse needs of researchers and varying intensities of studies.
- Fully equipped imaging department
MRI Magneton Lumina 3 Tesla – Siemens,
CT Somatom go.Up – Siemens, vEEG,
EMG, PSG, Holter T/ECG, Respiratory
poligraphy, Ultrasound – for
cardiovascular and soft tissue
imaging.
- Outpatient clinic system
- Day hospital for extended monitoring periods
- Interdisciplinary approach
- On site/local availability of modern integrated diagnostic
- Full time medical & project teams
World-class patient engagement
Enroll the right patients
faster
Neuroaxis
Research accurately forecasts and meets
enrollment and retention targets,
including for high-volume and
hard-to-recruit research programs. Our
growing patient pool covers all fields
of neurological conditions (including
rare diseases) and is expanding toward
cardiology, psychiatry, rheumatology,
and other medical fields. We nourish an
excellent relationship with our current
patients and have the means to bring in
any type of patient your study may
need.
- Access to proprietary, physician, and practice databases
- Immediate enrollment support upon study starts
- Intensive community outreach
- Participant adherence and retention programs
- Access to niche populations, procedures, and facilities


Investigators you can trust
Experienced Site Staff
Team
Our reputation for
a well-trained, enthusiastic, and
high-quality workforce is nationally
recognized, with accessible experienced
principal investigators. Neurology
studies will be led by renowned
neurologist doctor Dan Mitrea. A
seasoned site staff with a wealth of
expertise ensures the highest standards
of quality and efficiency in every phase
of your research journey.
- Doctors with international experience, committed to research
- Rare disease specialists
- Integrated procedures
- Project management/study coordination
- Scientific advice for protocol development
Clinical trial proficiency
Expertise that meets your every
need
Our experience
with clinical studies and regulatory
guidelines allows us to provide advice
on the most appropriate way to generate
robust evidence through your clinical
trial. Our expert team ensures
scientific advice for protocol
development in every stage of the study,
to support the timely and sound
development of high-quality and
effective results, for the benefit of
patients.
- Professional data collection and management
- Adherence to Good Clinical Practice (GCP)
- Ethical Considerations
- Safety Monitoring
- Adaptability


Fast activation
Streamlined start-up process
With over 6 years of
experience in project management,
Neuroaxis focuses on helping you
expedite your site selection process by
supplying fast, accurate responses to
questionnaires and feasibility studies.
We offer competitive fully executed
timelines and our straightforward
regulatory process, and standardized
contract agreement reduce start-up time
by up to two weeks from your first call.
- Collaboration and Communication
- Realistic Timelines ≤30 days from study award to site initiation visit
- Clinical trial feasibility protocol
- Single point of contact ensures seamless communication
- Continuous process Improvement
Count on the experience of your investigators and research staff in all branches of neurology and more!
- Neuro-vascular
- Neuro-immunology
- Movement Disorders
- Somnology | Epileptology
- Neuro-oncology
- Neurodegenerative
- Diseases
- Rare Neurological Diseases
- Craniofacial Pain
- Conditions of the Peripheral Nervous System
- Neuro-infections
- Inherited and Acquired Metabolic Disorders with Neurological Impact
- Neuroimaging
- Doppler Ultrasound
- Electromyography (EMG)
- Anxiety Disorder
- Depression
- Personality Disorders
- Sleep Disorders
Let’s talk about
your next trial!
- cercetare@neuroaxis.ro
- +40 722 734 213
- Bucharest, Calea Șerban Vodă nr. 206, 1st Floor